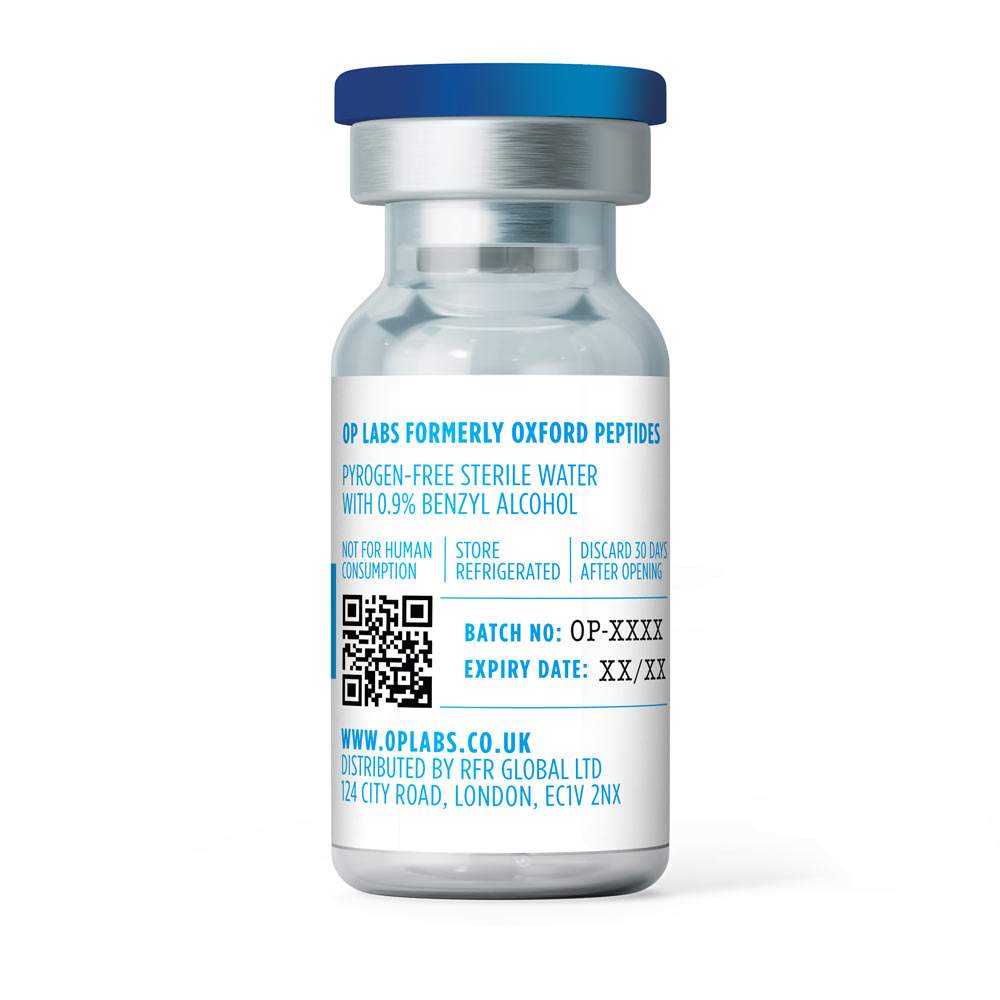
- OP Labs formerly Oxford Peptides
- Batch manufactured to specification
- Store at controlled room temperature unless otherwise specified
- Sold for research purposes only
- Contact us for Wholesale Orders
Download COA here: Bacteriostatic_Water_COA.pdf
Please note: if you have a different Batch ID, please contact us for the latest COA.
Bacteriostatic Water
Synonyms / Designations: Bacteriostatic Water for Injection (research use) Sterile Water with Benzyl Alcohol
CAS Number: 7732-18-5 (water) 100-51-6 (benzyl alcohol component)
Molecular Formula: H2O (with 0.9% benzyl alcohol, w/v)
Molar Mass 18.02 g/mol (water)
Composition: Sterile water for injection containing 0.9% benzyl alcohol as a bacteriostatic preservative
Product Classification Sterile aqueous diluent; bacteriostatic solvent
Purity: Manufactured to pharmacopeial specification (USP/Ph. Eur. grade components)
Appearance: Clear, colourless, sterile solution
Pack Size: 10 mL
Storage: Store in a cool dark place. Discard 30 DAYS after opening.
Solubility: Fully miscible with water and aqueous buffers
Description & Mechanism
Bacteriostatic Water is a sterile aqueous solution containing a low concentration of benzyl alcohol, formulated to inhibit microbial growth during repeated vial access. It is widely used in laboratory, pharmaceutical, and formulation research as a diluent for the reconstitution and dilution of injectable-grade compounds under controlled experimental conditions.
In biochemical and cellular research systems, Bacteriostatic Water is employed to evaluate compound solubility, stability following reconstitution, and short-term storage behaviour. The presence of benzyl alcohol provides bacteriostatic activity by disrupting microbial cell membranes, thereby reducing contamination risk during multi-use experimental workflows.
Applications in Research
- As a standard diluent for peptide and protein reconstitution
- Preparation of injectable-grade research compounds for in vitro or ex vivo studies
- Investigation of peptide and biologic stability following reconstitution
- Formulation testing and solvent compatibility studies
- Multi-use experimental workflows requiring bacteriostatic protection
Specifications Summary
Purity: Manufactured to specification
Appearance: Clear, colourless sterile solution
Solvent Type: Bacteriostatic aqueous diluent
Preservative: Benzyl alcohol 0.9% (w/v)
Solubility: Fully miscible with water and aqueous buffers
Storage: Controlled room temperature
Pack Size: 10 ml
Handling, Use & Stability
- Use aseptic technique when accessing vial contents
- Avoid contact with non-sterile surfaces
- Do not use if solution becomes cloudy or discoloured
- Seal vial promptly after use to maintain sterility
- Avoid exposure to excessive heat or prolonged light
- Do not freeze
Precautions & Notes
- Not all peptides or biologics are compatible with benzyl alcohol; compatibility should be assessed experimentally
- Buffer composition, compound sensitivity, and storage duration may influence stability
- Not suitable for neonatal or in vivo clinical use
- Intended strictly for laboratory research use
References
European Pharmacopoeia. Sterile Water and preserved preparations monographs.
https://www.edqm.eu/en/european-pharmacopoeia
Rowe R.C., Sheskey P.J., Quinn M.E. Handbook of Pharmaceutical Excipients. Pharmaceutical Press.
https://www.pharmpress.com/product/9780857110275/handbook-of-pharmaceutical-excipients
Trissel L.A. Handbook on Injectable Drugs. American Society of Health-System Pharmacists.
https://www.ashp.org/products-and-services/ashp-publications/handbook-on-injectable-drugs
Keywords: Bacteriostatic Water, Sterile Water, Benzyl Alcohol Solution, Research Diluent, Peptide Reconstitution, Laboratory Solvent